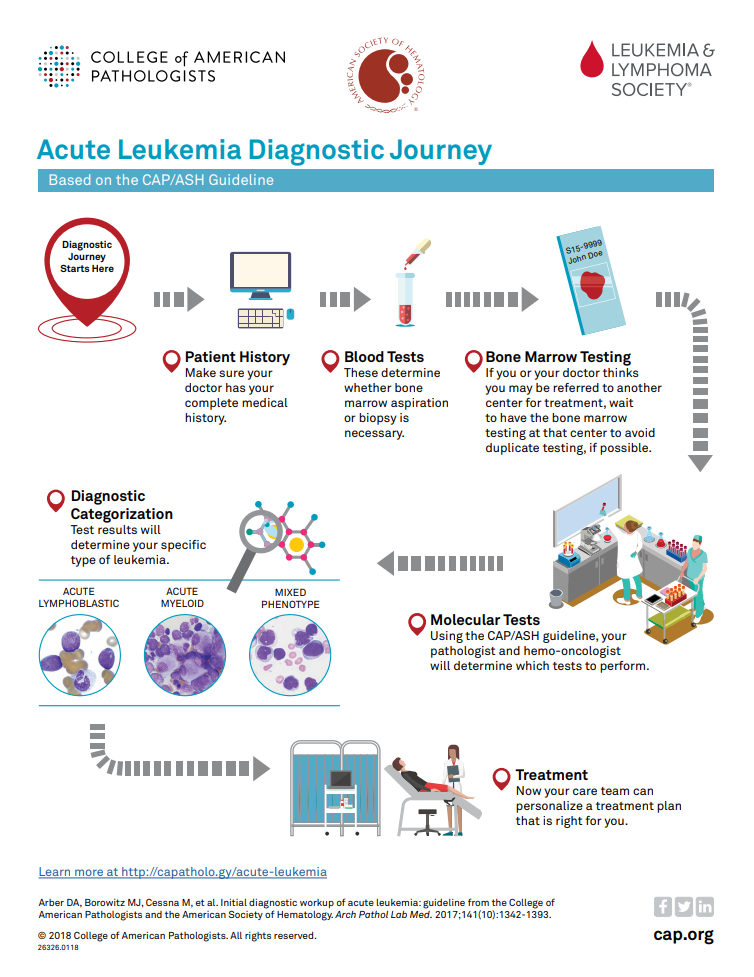
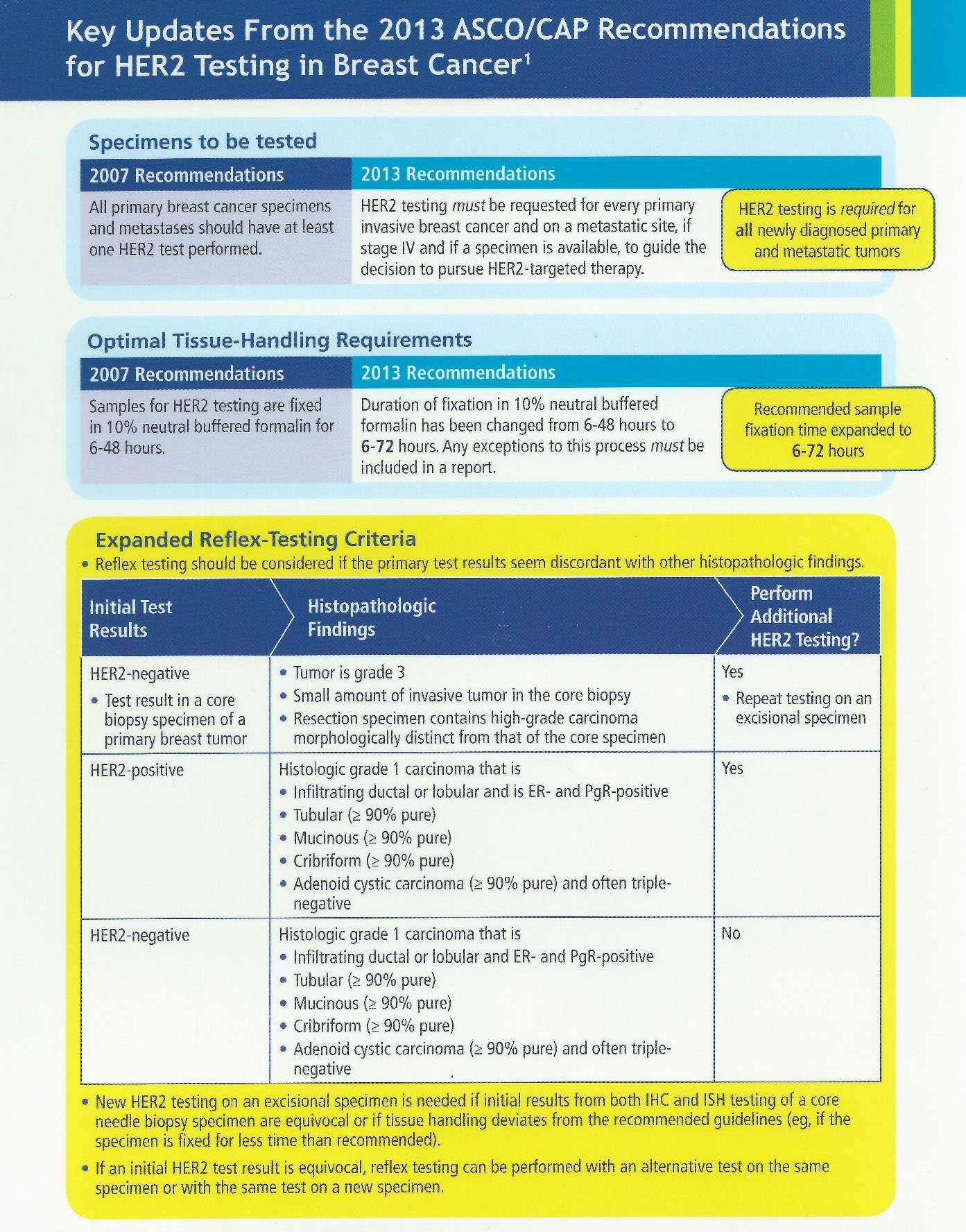
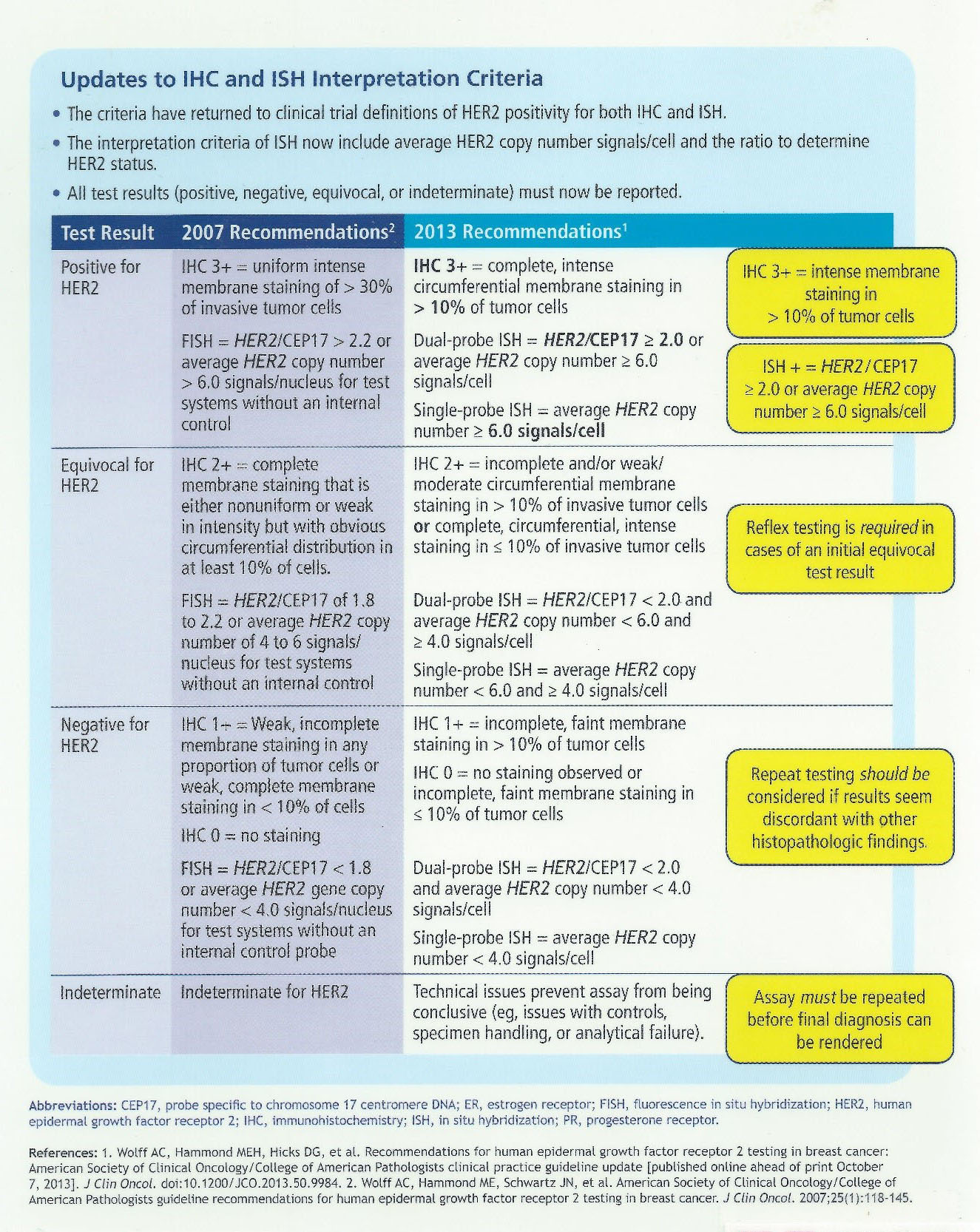
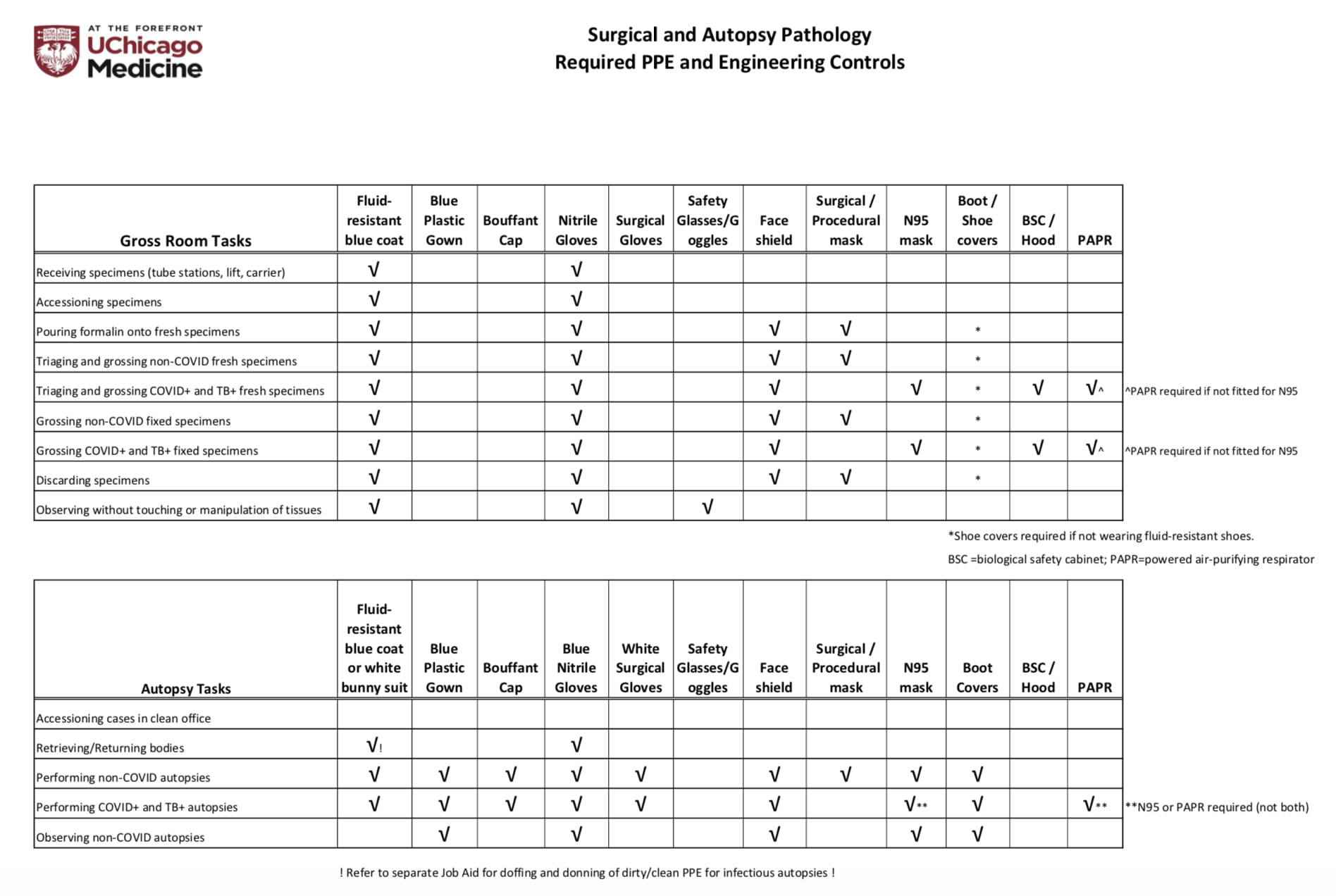
Fragment of CAP cancer checklist and ADASP guidelines for reporting... | Download Scientific Diagram

Medicina | Free Full-Text | Awareness of Placental Pathologic Examination Criteria and Utilization of Pathology Reports among Obstetricians
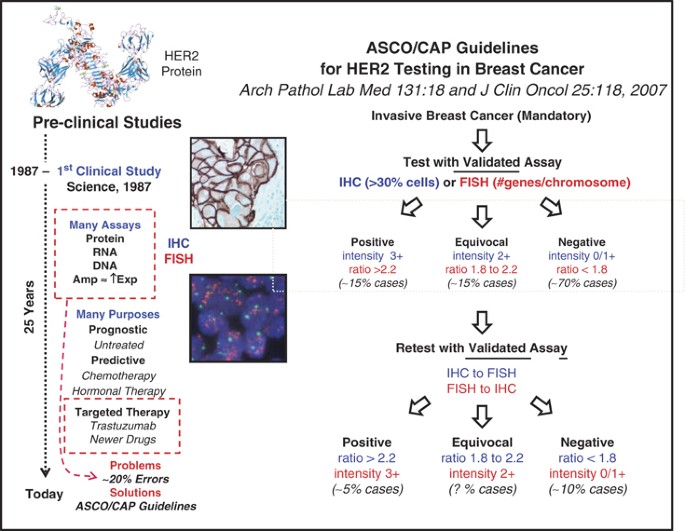
Issues and updates: evaluating estrogen receptor-α, progesterone receptor, and HER2 in breast cancer | Modern Pathology

Estrogen and Progesterone Receptor Testing in Breast Cancer: ASCO/CAP Guideline Update | Journal of Clinical Oncology
Page 1 of 6 College of American Pathologists - Labs seeing the light on practice guidelines 9/20/2010 http://www.cap.org/apps/ca
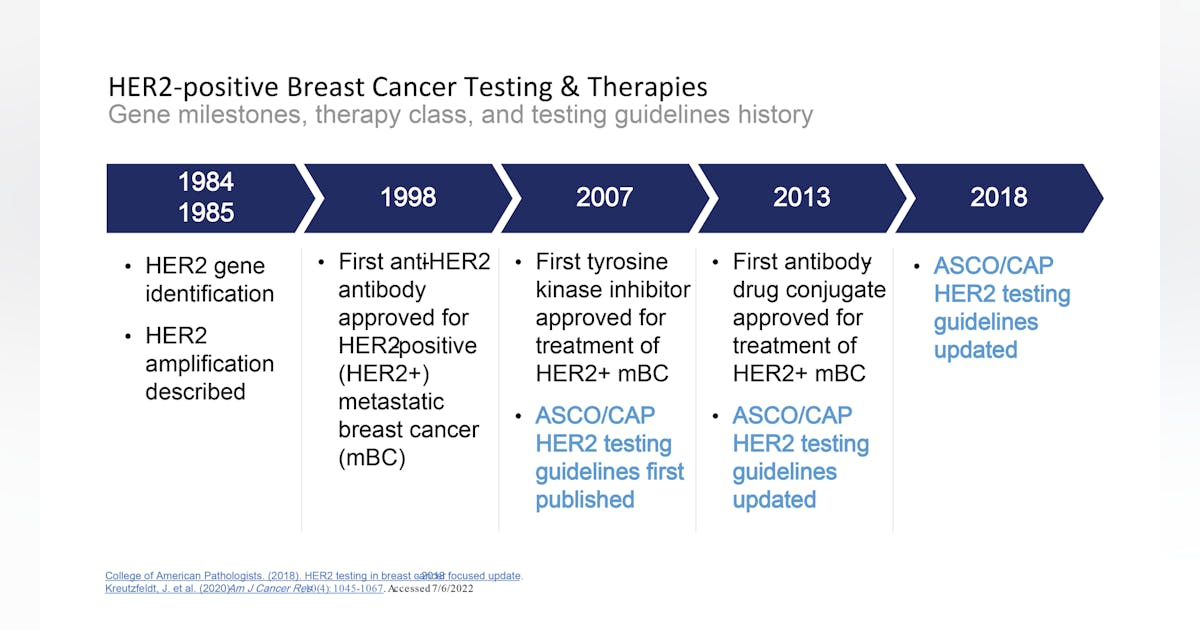
Breast cancer biomarkers, and a new clinical category for HER2 expression | Medical Laboratory Observer
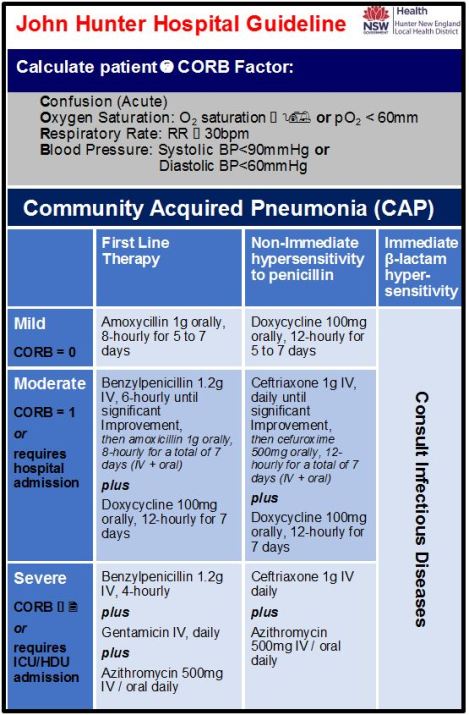
Why does our local CAP guideline differ from Australian Therapeutic Guidelines? | AIMED - Let's talk about antibiotics

PDF) American Society of Clinical Oncology/College of American Pathologists Guideline Recommendations for Immunohistochemical Testing of Estrogen and Progesterone Receptors in Breast Cancer
DATE: TO: Reference Lab Clients SUBJECT: CAP-NSH Uniform Labeling Requirements for Blocks and Slides in Surgical Pathology The C